Why the most consequential number in pharma is the one nobody says out loud
Ninety-nine of every hundred milligrams in the most consequential drug launch of the decade are paid for, swallowed, and never used. That is not an outlier. It is the architecture.
(11-minute read)
The Number on the Label
On the FDA label for the most successful pharmaceutical launch of the decade, there is a number printed in plain text. Most readers skip past it. Most clinicians do not internalise it. Most patients have never been told.
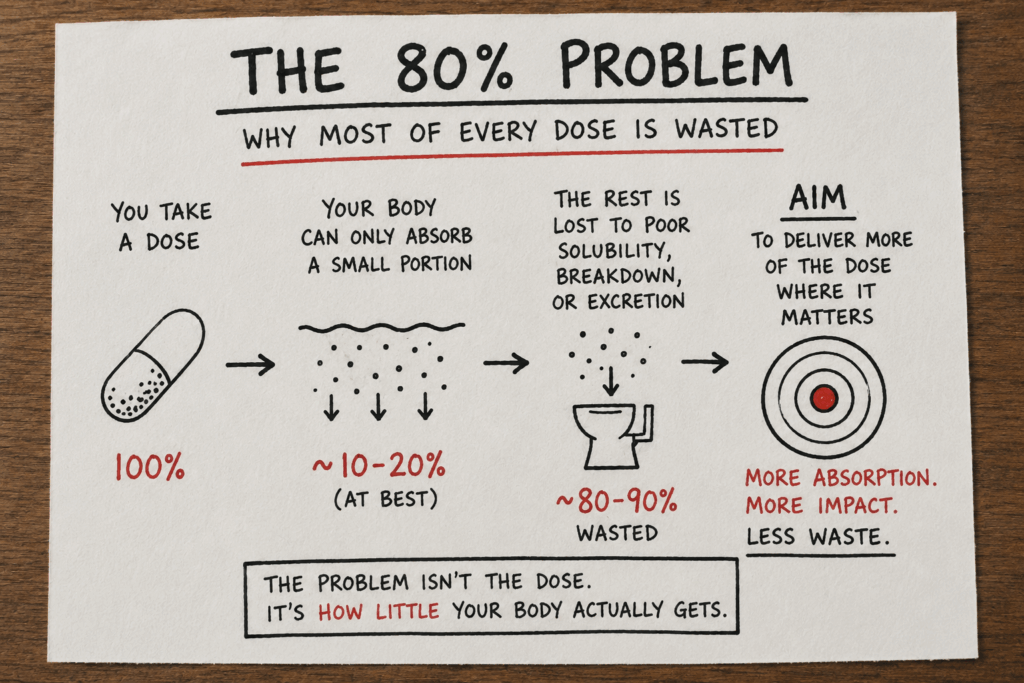
The number is 0.4 to 1.
That is the absolute bioavailability of oral semaglutide — Rybelsus, the first orally bioavailable GLP-1 — measured in percent.
For every 100 milligrams a patient swallows, the bloodstream sees less than one milligram. The other ninety-nine are paid for, swallowed, metabolised, excreted — and never used.
This is not a fringe case. It is the most consequential pharmaceutical launch in modern history. And it is approved, prescribed, paid for, and reimbursed at a one-percent yield.
If any other industry shipped a product at a one-percent yield, the industry would be replaced. Pharma has built a billion-dollar franchise on it.
This is the 80% Problem. Not a single drug. Not a single failure. The structural reality that, for most oral medicines, most of every dose never does any therapeutic work — and that the cost of that loss is paid by the patient and the planet, not the manufacturer.
Where the Dose Disappears
Between the swallow and the bloodstream, an oral drug runs a gauntlet:
- Disintegration and dissolution — the tablet must break apart and dissolve at the right place, at the right rate.
- Gastric degradation — stomach acid destroys a fraction.
- Limited absorption — only a portion crosses the intestinal wall.
- First-pass metabolism — the liver removes more before the drug ever reaches its target tissue.
Each step strips quantity. Each step adds variability.
For some molecules, what reaches systemic circulation is a respectable 60–80%. For many, it is 20–40%. For oral semaglutide, as we have just seen, it is below one percent. The number is not a curiosity. It is the entire economic and clinical model of the drug.
The Compensation Trap
When a drug fails to do enough, pharma has three responses, in order of preference.
- Increase the dose.
- Increase the frequency.
- Tolerate variability by widening the therapeutic window.
Each is reasonable in isolation. Together, they form a trap.
Because here is the asymmetry no-one talks about.
— · —
The swallowed dose is the safety dose.
The absorbed dose is the medicine.
— · —
Therapeutic effect scales with what arrives at the target. Side-effect burden scales with what was swallowed. They are different denominators, and the industry has built a hundred-year safety profile around the wrong one.
When you raise the swallowed dose to recover therapeutic effect, you do not just buy back the missing 80%. You also pay the full safety penalty of the swallowed mass — twice. Once on what was wasted. Once on what arrived.
The wasted dose is not biologically silent. It still hits the stomach. It still passes through the liver. It still competes for transporters, displaces other drugs at metabolic enzymes, irritates mucosa, generates reactive metabolites. The “lost” 80% is only lost from a therapeutic standpoint. From a toxicity standpoint, the patient swallowed all of it.
This is the compensation trap.
We respond to drug loss by adding the safety burden of more drug loss.
The Yield Problem Every Other Industry Solved
Almost every other industry has rebuilt itself around its yield problem.
James Watt’s separate condenser, patented in 1769, transformed steam-engine efficiency and, with it, the energetic foundation of the industrial revolution. Sadi Carnot’s 1824 work on heat-engine theory gave engineers the upper bound. A century of engineering then closed the gap toward it.
Coal-fired power stations operated at around 33% thermal efficiency for decades. Engineers spent a hundred years on supercritical steam, combined-cycle generation, cogeneration — and the world ultimately abandoned coal entirely. Two-thirds of the input lost as heat was no longer acceptable.
Internal-combustion engines climbed from roughly 12% efficiency in the Model T era to about 40% in modern Atkinson-cycle hybrids. Electrification is now finishing the job.
Modern turbofan jet engines convert about 40% of fuel energy to thrust. Aerospace has spent seventy years moving that number a single percentage point at a time.
— · —
Pharma has no James Watt.
We have had no separate condenser in two centuries.
— · —
For most oral drugs, bioavailability sits between 20% and 40%. For some, in single digits. The number has not moved meaningfully in fifty years.
The analogy is imperfect — biological systems vary in ways heat engines do not. But the principle holds. When an industry leaves a major efficiency gap untouched for decades, the explanation is rarely science. It is incentive.
The Bill Is Paid in Hospital Beds
The cost is countable, and it is paid in hospital beds.
NSAIDs, the workhorses of pain medicine, are absorbed well — but their gastric exposure is proportional to the swallowed dose. Approximately 107,000 NSAID-related gastrointestinal hospitalisations occur in the United States every year, with mortality estimates ranging from 3,200 to 16,500 deaths annually.
Paracetamol is the most common cause of acute liver failure in the United States, accounting for roughly 50% of all cases — 56,000 emergency department visits, 2,600 hospitalisations, and about 500 deaths every year. Half of those poisonings are unintentional. Patients stacking products. Missing instructions. Taking “a little more” because the last dose did not work.
The pharmacology that made these drugs cheap is the pharmacology that makes them dangerous when dosed up to compensate for inconsistent relief.
Multiply that pattern across every oral therapy that has ever been dose-escalated for inadequate response — statins, anticoagulants, antidepressants, opioids, immunosuppressants — and a pattern emerges that is not a sequence of unlucky individuals.
It is an architecture.
And in Tonnes of Solvent We Will Never Recover
There is a second cost, paid before the dose ever reaches the patient.
Active pharmaceutical ingredients are extraordinarily energy-intensive to make. The carbon footprint of API synthesis runs from 50 to 1,000 kg of CO₂ per kilogram of API — two to fifty times the footprint of speciality chemicals. The process mass intensity — the kilograms of raw material consumed per kilogram of finished API — sits between 70 and 433, with solvents alone accounting for half. Synthesis yields commonly run 30–60%, dropping to 5–10% for complex molecules.
Stack the losses end-to-end. Synthesis loses 40–95% of starting material. Formulation loses single-digit percentages more. The patient swallows what remains. Their gut absorbs 20–40% of what they swallowed.
The mass that ends up doing therapeutic work is, for many drugs, less than one percent of what was originally synthesised.
— · —
The carbon was real.
The yield, in clinical terms, was zero.
— · —
We are mining solvents and hydrocarbons to manufacture API that, for a substantial share of every dose, is never delivered, never used, and never benefits a patient.
The Three Penalties
Put the asymmetry plainly. For a typical oral drug, three things happen with every dose:
- Therapeutic benefit is paid for by the absorbed fraction.
- Safety burden is paid in full by the swallowed dose.
- Manufacturing and environmental burden is paid in full by the manufactured dose — which is larger again than the swallowed dose, because of formulation losses, process losses and supply-chain shrinkage.
Two of the three penalties are paid in full for the wasted 80%. Only one
— · —
The next era of pharma will not be defined by what we synthesise, what we compress, or what we prescribe.
It will be defined by what we no longer waste.
How much of this drug actually reaches the body — and how much of the rest is paid in side effects, in carbon, and in trust?
Everything else is bookkeeping.
Ibumix is a UK-based pharmaceutical platform company developing new liquid drug delivery platforms — designing oral medicines around what the body receives, not what the factory produces.
