Why Most Drugs Fail Before They Even Start Working

There is a moment in every oral drug’s life that determines everything.
It happens silently.
Invisibly.
Before the drug ever reaches the bloodstream.
It is the moment the solid must become a solution.
If a drug does not dissolve, it does not exist—at least not to the body.
This is the dissolution trap.
And most modern drugs fall into it.
1. The Assumption That Breaks Everything
The entire concept of a tablet or capsule rests on a simple assumption:
That once swallowed, the drug will dissolve.
It feels reasonable.
Almost trivial.
But in reality, dissolution is one of the most fragile steps in drug delivery.
Because the human body is not a controlled environment.
It is:
- Variable
- Reactive
- Chemically complex
And dissolution depends on all of it.
2. Solid to Solution: The Critical Transition
A drug in solid form cannot cross biological membranes.
To be absorbed, it must:
- Disintegrate (if in a tablet)
- Dissolve into gastrointestinal fluids
- Exist as free molecules in solution
Only then can it begin to move into the body.
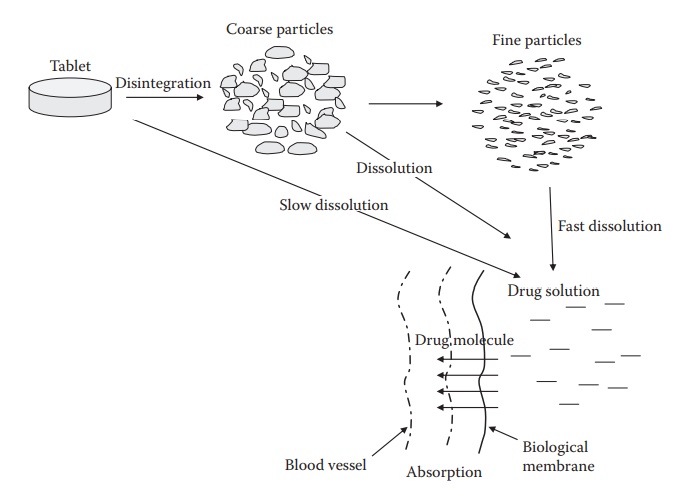
Every step introduces risk.
But dissolution is the narrowest gate.
It is the point where physics meets chemistry—and often fails.
3. The Solubility Problem
Many modern drugs are:
- Lipophilic
- Poorly water-soluble
- Chemically complex
Which means:
They do not naturally dissolve in the aqueous environment of the body
Instead, they:
- Clump
- Aggregate
- Remain partially undissolved
And undissolved drug is:
Pharmacologically invisible
It passes through the body without effect.
4. The Illusion of the Dose
When you take a 500 mg tablet, it feels like you are receiving 500 mg of drug.
But in reality:
- Only the dissolved fraction is available
- The rest is effectively lost
So the true delivered dose is not:
What you swallow
But:
What successfully dissolves
And that number is rarely constant.
5. Precipitation: Dissolution Reversed
Even when drugs do dissolve, the story doesn’t end there.
In some cases, they dissolve temporarily—
only to precipitate back into solid form.
This happens when:
- Concentration exceeds solubility limits
- Environmental conditions change (pH, bile, enzymes)
The result:
- A transient solution
- Followed by loss of availability
The drug appears… then disappears
This is one of the most overlooked failures in oral delivery.
6. The Body Is Not a Beaker
In the lab, dissolution is measured under controlled conditions:
- Constant pH
- Defined fluid volumes
- Controlled agitation
In the body, none of this holds.
Instead, dissolution is influenced by:
- Food intake
- Gastric emptying
- Intestinal motility
- Fluid variability
So the same tablet can:
- Dissolve well one day
- Poorly the next
Even in the same patient.
7. Why the Industry Works Around It
If dissolution is such a critical problem, why hasn’t it been solved?
Because the system has adapted—incrementally.
Through:
- Particle size reduction
- Salt formation
- Amorphous dispersions
- Surfactants and excipients
These approaches improve dissolution.
But they do not eliminate the dependency on it.
The system still relies on a fragile step—and tries to stabilise it
8. The Cost of the Trap
When dissolution is unreliable, the system compensates:
- Higher doses
- More complex formulations
- Increased variability
This leads to:
- Side effects (from excess dosing)
- Inconsistent efficacy
- Patient unpredictability
And ultimately:
A drug that works in theory—but not reliably in practice
9. The Deeper Insight
The dissolution trap reveals something fundamental:
We are forcing solid systems into liquid biology
Instead of designing drugs to exist in solution,
we design them as solids—and hope they convert.
That hope is where variability lives.
10. The Ibumix Perspective
Dissolution should not be:
- A bottleneck
- A variable
- A failure point
It should be:
Engineered out of the system
Because as long as medicine depends on:
- Disintegration
- Dissolution
- Environmental variability
It will never be fully controlled.
11. A Different Future
Imagine a system where:
- The drug is already in a bioavailable state
- No dissolution step is required
- Absorption begins immediately
Where:
- The body receives what is intended
- Not what happens to dissolve
In that world:
There is no trap—because there is no dependency
Final Line
Most drugs don’t fail at the target.
They fail in the transition from solid to solution.
And until that is solved,
medicine will remain an approximation.
