Why your ibuprofen isn’t working as well as it could
Peer-reviewed pharmacokinetic research has confirmed what formulation scientists have long suspected: the molecule is not the variable. The delivery system is. And the implications for how we design medicines are profound.
Every day, millions of people reach for an ibuprofen tablet expecting the same result. They assume that a 400 mg tablet is a 400 mg tablet — that the dose is what matters, and the rest is packaging.
The science disagrees. Fundamentally.
A significant body of pharmacokinetic research, including a rigorously conducted study published in European Journal of Pharmaceutics and Biopharmaceutics, has demonstrated that different formulations of identical ibuprofen doses can produce meaningfully different clinical outcomes. The same molecule. The same amount. Dramatically different absorption profiles, onset times, and therapeutic durations.
This is not a marginal effect. It is the central challenge of pharmaceutical delivery — and one that the industry has historically underestimated.
The molecule is not the limiting factor. The question has never been what we put in the tablet. It has always been how much of it actually reaches the patient.
The therapeutic window: what the numbers actually mean
To understand why formulation matters so much, you first need to understand how ibuprofen actually works in the body — and how little room for error there is.
Ibuprofen is only effective when plasma concentrations exceed specific thresholds. Research has established two critical values: a minimum concentration of 6.8 µg/mL, below which the drug produces little meaningful clinical benefit, and a higher threshold of 10.1 µg/mL, above which stronger pain and fever relief is achieved.
Every formulation decision — tablet type, dissolution rate, excipient chemistry — determines how quickly those thresholds are reached, how long they are maintained, and how much of the dose is simply lost.

Standard immediate-release tablets are competent but imperfect. They absorb adequately under typical conditions, but absorption can be delayed by food, gut motility differences, and formulation quality. For many patients, a meaningful portion of each dose never crosses the therapeutic threshold at the right time — or sustains it for long enough.
Not all tablets are created equal
The published research compared four distinct formulations of ibuprofen: a standard immediate-release 400 mg tablet, a standard 200 mg tablet, a lysinate salt formulation equivalent to 400 mg, and a novel bi-layer tablet combining both immediate- and sustained-release components.
The findings were instructive.
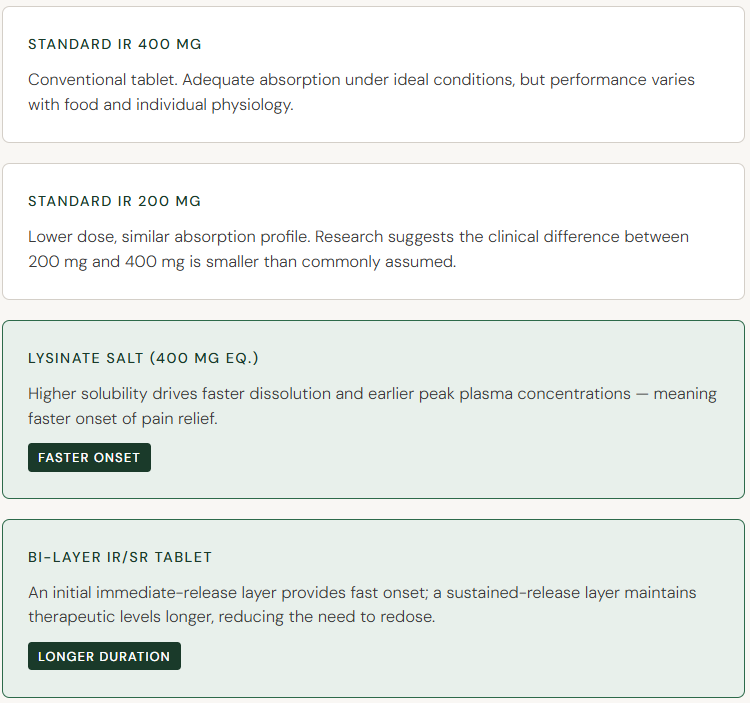
The lysinate salt form reached therapeutic plasma concentrations faster than the standard tablet — not because of more drug, but because of how the drug was presented to the body. The bi-layer formulation extended the therapeutic window significantly, providing coverage that a single immediate-release dose simply cannot.
These are not trivial differences. For a patient managing post-operative pain, fever in a child, or chronic inflammation, the difference between peak concentration at 30 minutes versus 60 minutes is clinically meaningful.
The dose myth: more is not the answer
Perhaps the most counterintuitive finding in this area of pharmacokinetic research concerns the relationship between dose and effect. Ibuprofen has a relatively flat dose-response curve — meaning that as plasma concentrations rise, the incremental gain in clinical benefit diminishes significantly.

A 30% increase in circulating drug produces less than a 10% improvement in clinical outcome. The implication is clear and important: escalating dose is a crude and inefficient approach to improving ibuprofen’s performance. The more elegant — and the more scientifically grounded — solution is to ensure that the dose you are already taking is absorbed and maintained as efficiently as possible.
This reframes the entire innovation opportunity. The goal is not higher doses. It is smarter delivery.
The role of computational modelling
One of the study’s methodological contributions deserves particular attention: the validation of physiologically based pharmacokinetic (PBPK) modelling as a reliable tool for predicting real-world clinical outcomes.
By constructing detailed computational models of how ibuprofen moves through the body — accounting for absorption kinetics, plasma binding, gut physiology, and individual variability — researchers were able to simulate clinical trials in virtual populations before conducting them in humans. When those predictions were compared against actual trial data, the correlation was high: R² values of 0.8 or above across key pharmacokinetic and pharmacodynamic measures.
This matters because it validates a development philosophy. Formulation science no longer needs to rely solely on costly, time-consuming empirical testing. Predictive modelling, when built on rigorous mechanistic foundations, can guide development intelligently — identifying the most promising formulation candidates before a single human trial is run.
Delivery is the next frontier of pharmaceutical innovation
The research described above confirms what Ibumix was founded on: that the active ingredient is not the limiting factor in most oral medicines. The constraint is delivery — how efficiently the molecule is absorbed, how reliably it crosses the therapeutic threshold, and how consistently it performs across the full diversity of human physiology.
Ibumix is engineering liquid crystal-based excipient systems designed to address this constraint directly. Using amphiphile-driven self-assembly — creating tunable lamellar, hexagonal, and cubic phase structures — the platform is built to enhance solubility, improve absorption kinetics, and reduce formulation variability.
Ibuprofen and naproxen are the starting points. The platform extends far beyond them. If the pharmacokinetic research tells us anything, it is that the opportunity is not in finding new molecules. It is in making every molecule perform at its actual potential.
What this means for patients — and for pharmaceutical development
For the patient reaching for an OTC painkiller, the practical takeaway is simple: the form of ibuprofen you take matters. A lysinate salt formulation will typically act faster than a standard tablet. A sustained-release formulation will last longer. Understanding this allows for more informed choices about which product is appropriate for a given situation — fast-onset pain relief versus sustained fever management, for example.
For pharmaceutical developers, the implications run deeper. Formulation decisions are clinical decisions. Excipient selection, dissolution profile, and delivery architecture are not downstream manufacturing considerations — they are core determinants of whether a medicine works as intended.
The published research validates this position with rigorous, peer-reviewed data. The question it leaves open — and the question Ibumix exists to answer — is what becomes possible when you engineer the delivery system from first principles, rather than accepting the constraints of existing excipient technology.
The molecule has always been only half the story. Ibumix is here to finish it.
Primary source: “Optimizing ibuprofen dosing: insights from in vivo and virtual pharmacokinetic trials,” European Journal of Pharmaceutics and Biopharmaceutics, February 2026. Published via ScienceDirect. Additional pharmacokinetic and pharmacodynamic data referenced from peer-reviewed literature on PBPK/PD modelling and ibuprofen dose-response relationships. Link to article: Optimizing ibuprofen dosing: insights from in vivo and virtual pharmacokinetic trials – ScienceDirect
Disclaimer: This article is intended for informational and educational purposes. It does not constitute medical advice. Always follow dosing instructions provided with any medicine and consult a qualified healthcare professional where appropriate.
